AJOVY Trademark of TEVA PHARMACEUTICALS INTERNATIONAL GMBH - Registration Number 5645449 - Serial Number 87902122 :: Justia Trademarks

Teva Announces Positive Top-Line Results from AJOVY® (fremanezumab) Clinical Trials in Japan | Business Wire
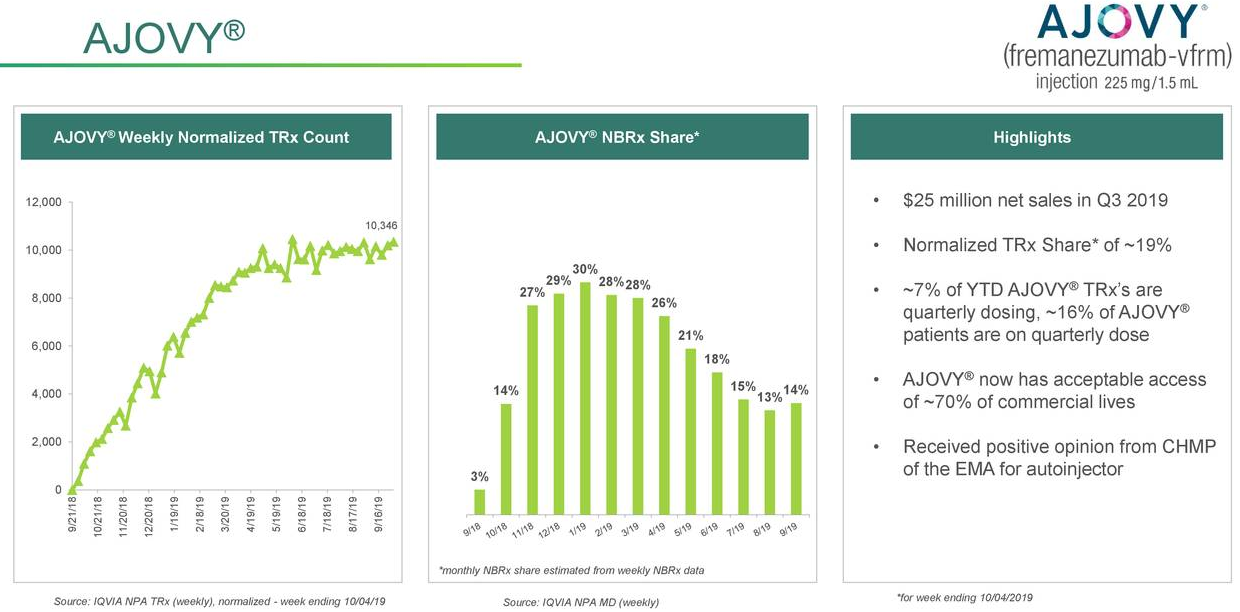
Teva taps new North American commercial chief as key meds Ajovy, Austedo lag their rivals - Opera News
Ajovy auoinjector with box and patient instruction booklet on a table. Ajovy is one of four newly FDA approved migraine preventatives Stock Photo - Alamy
Teva Pharmaceutical Industries : Announces U.S. Launch of Autoinjector for AJOVY® (fremanezumab-vfrm) Injection | MarketScreener

Teva Canada Announces New Autoinjector for AJOVY® for the Preventive Treatment of Migraine in Adults | Business Wire